METEOROID Clinical Trial
Sponsored by Hoffmann-La Roche
Learn more about the study from the Meteoroid website. (source https://forpatients.roche.com/).
UPDATED January 29, 2026
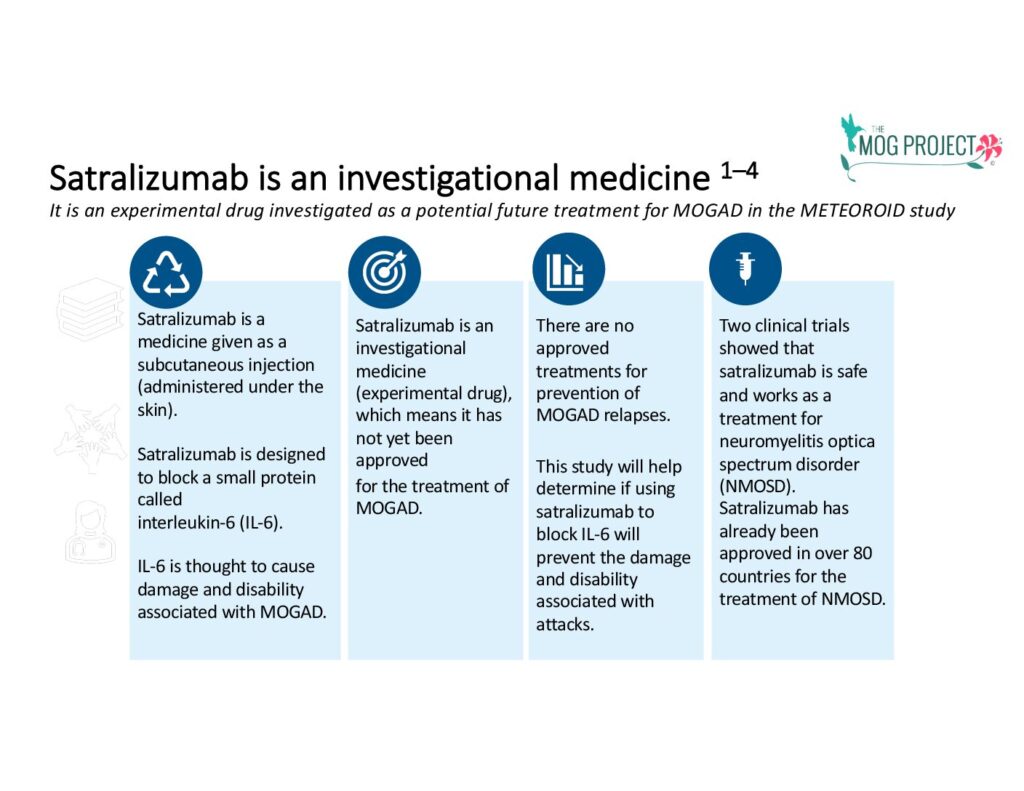
Hoffmann-La Roche/Genentech has announced positive results from a 44-month randomized double-blind place-controlled Phase III study—the METEOROID clinical trial—which studied satralizumab in MOGAD. This marks the first successful Phase III clinical trial in MOGAD. The goal of the METEOROID study was to confirm satralizumab’s significant effectiveness in preventing and/or delaying the time to MOGAD relapses, particularly in individuals with recurrent disease. The trial met its primary endpoint, achieved key secondary endpoints, and affirmed a positive safety profile.
Satralizumab blocks the Interleukin-6 (IL-6) receptor which prevents it from causing inflammation and aims to prevent future attacks or relapses. There are no approved medications for MOGAD, so the announcement today has been long awaited by a global audience of patients with MOGAD. This same therapy was previously approved by the U.S. Food and Drug Administration (FDA) and other international authorities for Neuromyelitis Optic Spectrum Disorder (NMOSD), a different autoimmune disease of the central nervous system. Interleukin-6 is involved in the pathogenesis of NMOSD.
Julie Lefelar, Founding Executive Director, and Amy Ednie, Founding President of The MOG Project, the pre-eminent global nonprofit organization wholly dedicated to MOGAD, issued the following statement:
“For children and adults with MOGAD and their loved ones, this is an historic day! With no approved treatments for MOGAD, the news today indicates that it is possible to develop safe and effective therapies for MOGAD. We are greatly encouraged by the positive top-line data which shows the 44- month PHASE III trial met its primary endpoint, i.e., to prevent and/or delay the time to MOGAD relapses. These results provide hope that this complex neuroimmune disease can be brought under control and that, eventually, a cure can be found for the hundreds of thousands of people living with MOGAD worldwide."
"On behalf of the global community of patients and family members, we applaud Hoffmann-La Roche/Genentech’s landmark achievement and their investment in finding new, safe, and effective treatments for MOGAD. And we salute the heroic patients in the trial whose participation has helped not only themselves but the larger community of patients who have long awaited this day.”
With gratitude,
The MOG Project Team
The MOG Project (TMP) is the preeminent global nonprofit voluntary health organization wholly dedicated to finding the causes of and cure for MOGAD, and providing support, services, and hope to all people affected by MOGAD. Founded in 2018, TMP has a global network of researchers, scientists, international Regional Delegates, and support groups and conducts programs of research, education, and advocacy.
Watch This video to help you understand the METEOROID Trial
The video below provides a great review of what Satralizumab does and what to expect when you enter a clinical trial. The slides presented in the video are available below as well and include a downloadable, accessible PDF version that you can also print and take to your doctor.
Michael on Meteoroid for MOGAD: Understanding the Satralizumab Clinical Trial
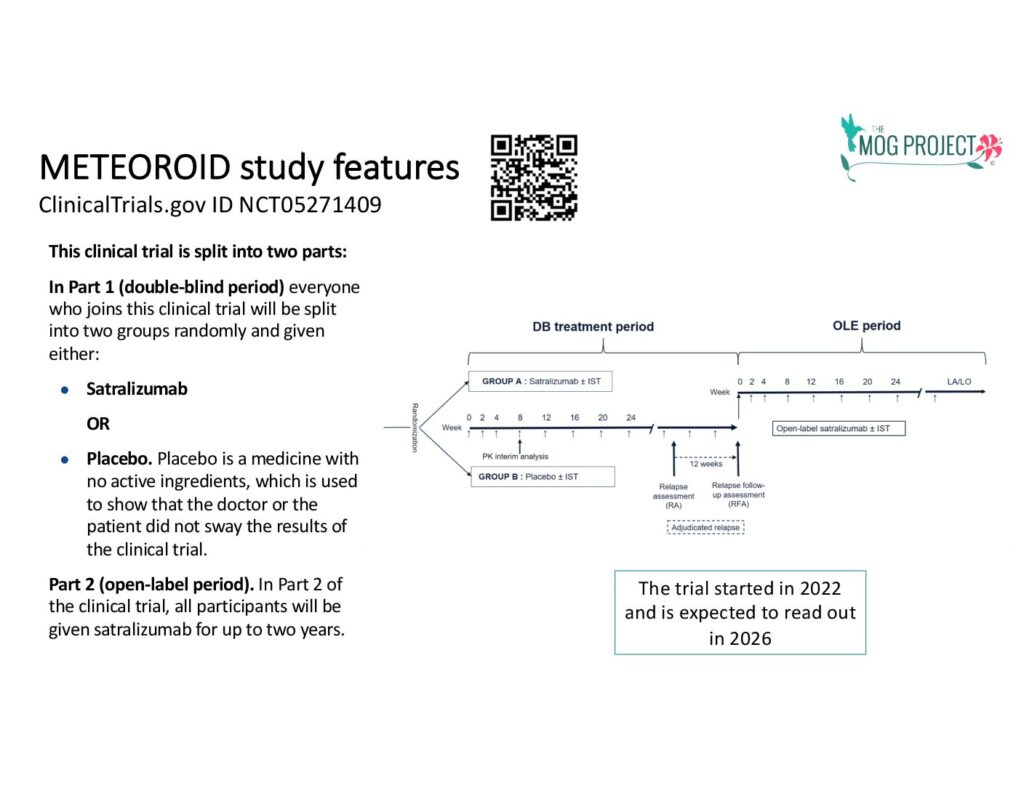
The infographics below will help you understand the process in this trial courtesy of https://forpatients.roche.com/: